Body centered Orthorhombic 9. A body-centered cubic unit cell structure consists of atoms arranged in a cube where each corner of the cube.
Face Centered Cubic Fcc Unit Cell Materials Science Engineering

Surface Analysis Of The Stress Induced Impurity Driven Face Centered Cubic Titanium Phase And The Ranging Lattice Parameter Sizes Sciencedirect

Body Centered Cubic 3d Model By Dr Gray S Models Matengswanu Fc8352c
Go to the general unit cell problems.
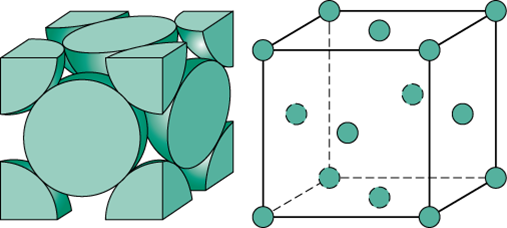
Face centered cubic. NaCl has a cubic unit cell. A face-centered cubic unit cell structure consists of atoms arranged in a cube where each corner of the cube has a fraction of an atom with six additional full atoms positioned at the center of each cube face. These are not always cube-shaped.
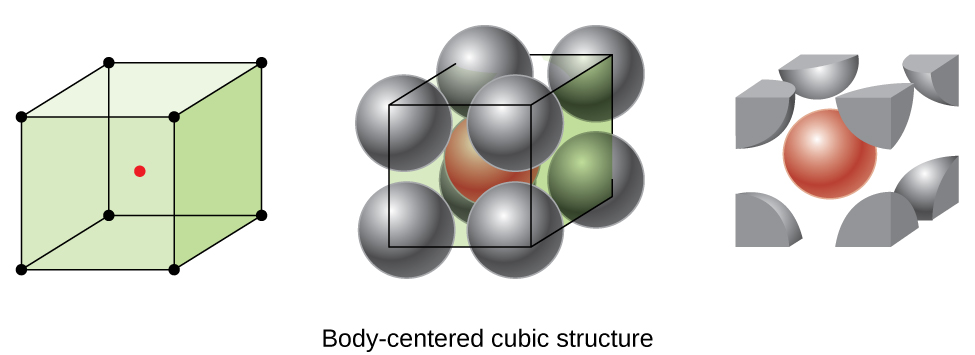
Body-centered cubic packing of anions is like cubic packing with an additional anion in each cubic hole. FCC is a close-packed structure with ABC-ABC stacking. What Does Body-Centered Cubic BCC Mean.
The cell looks the same whether you start with anions or cations on the corners. The Face-Centered Cubic FCC unit cell can be imagined as a cube with an atom on each corner and an atom on each face. As atoms of melted metal begin to pack together to form a crystal lattice at the freezing point groups of these atoms form tiny crystals.
We can think of this cell as being made by inserting another atom into each face of the simple cubic lattice - hence the face centered cubic name. In fact the presence of face-centered cubic unit cells in this structure explains why the structure is known as cubic closest-packed. The face-centered cubic structure can be seen in figure 3.
There are 8 corner positions and each position contributes one eighth to the unit cell. Unlike the simple cubic lattice it has an additional lattice point located in the center of the cube. Like tetragonal crystals except not square in cross-section when viewing the crystal on end these crystals form.
1 Calculate the average mass of one atom of Pd. Body-centered cubic BCC is the name given to a type of atom arrangement found in nature. At the same time the formation of body-centered cubic bcc and face-centered cubic fcc Ti phases are promoted.
FCC has 4 atoms per unit cell lattice constant a 2R2 Coordination Number CN 12 and Atomic Packing Factor APF 74. A face-centered cubic solid has atoms at the corners and as the name implies at the centers of the faces of its unit cells. Click here to buy a book photographic periodic table poster card deck or 3D print based on the images you see here.
This structure is also called cubic closest packing CCP. The edge length of the unit cell of Ta is 3306 pm. Atoms in an FCC arrangement are packed as closely together as possible with atoms occupying 74 of the volume.
The face-centered cubic unit cell is the simplest repeating unit in a cubic closest-packed structure. Body centered tetragonal 6. It is one of the most common structures for metals.
What Does Face-Centered Cubic FCC Mean. Lets take our simple cubic crystal structure of eight atoms from the last section and insert another atom in the center of the cube. Base centered monoclinic 12.
1 It seems like your. Return to the Liquids Solids menu. When heating iron above 912 C 1674 F its crystal structure changes to a face-centered cubic fcc crystalline structure.
Triclinic Cubic Tetragonal Orthorhombic Rhombohedral Hexagonal Monoclinic. It is best thought of as a face-centered cubic array of anions with an interpenetrating fcc cation lattice or vice-versa. Tantalum has a density of 1669 gcm 3.
The face-centered unit cell has 8 atoms at each corner and one in the center of each face. This new structure shown in the figure below is referred to as body-centered cubic since it has an atom centered in the body of the cube. Face centered Orthorhombic 10.
Body centered cubic 3. Face-centered cubic FCC or cF is the name given to a type of atom arrangement found in nature. Go to the face-centered cubic problems.
The smallest unit that when repeated gives the structure of the material is the unit cell. It is shown in the figure below. γ-iron can dissolve considerably more carbon as much as 204 by mass at 1146 CThis γ form of carbon saturation is exhibited in stainless steel.
The reason for the various colors is to help point out how the cells stack in the solid. It is one of the most common structures for metals. Its density is 12023 gcm 3Calculate the atomic radius of palladium.
10642 g mol 1 6022 x 10 23 atoms mol 1 1767187 x 10 22 gatom 2 Calculate the mass of the 4 palladium atoms in the face-centered cubic unit cell. Face centered cubic 4. Face Centered Cubic Figure 4.
This structure as well as the next structure we are going to discuss has the atoms packed as tightly as theoretically possible. These tiny crystals increase in size by the progressive addition of atoms. αβγ 90 a c a b c α β γ 90 β 90 αγ 90.
The face-centered cubic system is closely related to the hexagonal close packed hcp system where two systems differ only in the relative placements of their hexagonal layers. Youll also find octahedrons eight faces and dodecahedrons 10 faces. In a face centered cubic lattice atom A occupies the corner positions.
The Body-Centered Cubic BCC unit cell can be imagined as a cube with an atom on each corner and an atom in the cubes center. This crystal structure is known as face-centered cubic and has atoms at each corner of the cube and six atoms at each face of the cube. BCC has 2 atoms per unit cell lattice constant a 4R3 Coordination number CN 8 and Atomic Packing Factor APF 68.
There are six face centre positions. The plane of a face-centered cubic system is a hexagonal grid. Hence total number of A atoms per unit cell 8 1 8 1 Atom B occupied the face centre positions.
The study demonstrated that after short milling times between 2 h and 30 h the fraction of hexagonal close-packed hcp phase decreases. A calculate the mass of a tantalum atom. Base centered orthorhombic 8.
The resulting solid is not one. The real space and reciprocal space primitive translation vectors are. Besides the simple cubic sc and the face centered cubic fcc lattices there is another cubic Bravais lattice called body centered cubic bcc lattice.
Palladium crystallizes in a face-centered cubic unit cell. Unit cells for BCC and FCC. Each ion is 6-coordinate and has a local octahedral geometry.
Similar to cubic crystals but longer along one axis than the other these crystals forming double pyramids and prisms. In this form it is called gamma iron γ-Fe or Austenite. This means that there are 818 612.
The unit cell for cubic body-centered cubic and face-centered cubic are shown below. Face Centered Cubic fcc or Cubic Close Packed ccp These are two different names for the same lattice. Face-centered cubic is another name for cubic close packed.
The unit cell is body-centered cubic. Beginequation large veca_1fraca2hatxhatzquad veca_2fraca2hat. Cubic or Isometric.
Face Centered Cubic

The Face Centered Cubic Crystal Structure And The Theoretical Density Of Metals Youtube
1
10 6 Lattice Structures In Crystalline Solids Chemistry Libretexts

1 Surface Structure Of Ideal Face Centered Cubic Single Crystals Download Scientific Diagram
Face Centered Cubic Fcc Unit Cell Materials Science Engineering

Sketches Of Diamond Left Face Centered Cubic Center And Download Scientific Diagram
Question F3e7d Socratic
